How to Deduce the Alkali Metal When You Know the Anion Is Carbonate

Characteristic cleft pattern associated with the alkali–silica reaction affecting a concrete footstep barrier on a US state highway. Note the typical fatty attribute of the silicagel exudations imbibing the physical surface along the two sides of the cracks.[1] [2]
The alkali–silica reaction (ASR), more commonly known as "physical cancer", is a deleterious swelling reaction that occurs over fourth dimension in concrete betwixt the highly alkaline cement paste and the reactive amorphous (i.due east., not-crystalline) silica found in many common aggregates, given sufficient moisture.
This deleterious chemical reaction causes the expansion of the altered aggregate by the germination of a soluble and viscous gel of sodium silicate (Na2SiOiii · n HtwoO, likewise noted Na2H2SiO4 · n HtwoO, or N-South-H (sodium silicate hydrate), depending on the adopted convention). This hygroscopic gel swells and increases in volume when absorbing water: information technology exerts an expansive pressure inside the siliceous aggregate, causing spalling and loss of force of the concrete, finally leading to its failure.
ASR can atomic number 82 to serious cracking in concrete, resulting in disquisitional structural problems that can fifty-fifty force the demolition of a detail construction.[3] [four] [five] The expansion of concrete through reaction betwixt cement and aggregates was first studied by Thomas E. Stanton in California during the 1930s with his founding publication in 1940.[6]
Chemical science [edit]

Typical fissure pattern of the alkali-silica reaction (ASR). The gel exudations through the physical cracks have a characteristic yellow color and a high pH. The fatty attribute of the exudations imbibing the concrete porosity along the cracks is also a distinctive feature of ASR.
To attempt to simplify and to stylize a very complex ready of various reactions, the whole ASR reaction, after its complete evolution (ageing process) in the presence of sufficient Ca2+ cations available in solution, could be compared to the pozzolanic reaction which would exist catalysed past the undesirable presence of excessive concentrations of alkali hydroxides (NaOH and KOH) in the physical.[7] It is a mineral acrid-base of operations reaction betwixt NaOH or KOH, calcium hydroxide, also known as portlandite, or (Ca(OH)ii), and silicic acid (H4SiO4, or Si(OH)4). For simplifying, after a consummate substitution of the brine cations with the calcium ions released past portlandite, the brine-silica reaction in its ultimate stage leading to calcium silicate hydrate (C-S-H) could be schematically represented as following:
- Ca(OH)ii + H4SiO4 → Caii+ + H2SiO4 2− + two H2O → CaHiiSiOiv · 2 HtwoO
Hither, the silicic acid H4SiO4, or Si(OH)iv, which is equivalent to SiO2 · 2 H2O represents hydrous or baggy silica for the sake of simplicity in aqueous chemistry.
Indeed, the term silicic acid has traditionally been used as a synonym for silica, SiO2. Strictly speaking, silica is the anhydride of orthosilicic acid, Si(OH)4.
- SiO2↓ + two H2O ⇌ Si(OH)iv
An ancient industrial notation referring to H
two SiO
3 , metasilicic acid, is also often used to depict the alkali-silica reaction. However, the metasilicic acrid, H
two SiO
three , or SiO(OH)
2 , is a hypothetic molecule which has never been observed, fifty-fifty in extreme diluted solutions because H
2 SiO
3 is unstable and continue to hydrate.
Indeed, contrary to the hydration of CO2 which consumes just one h2o molecule and stops at H2CO3, the hydration of SiO2 consumes ii water molecules and continues one step further to course H4SiO4. The departure in hydration behaviour between SiO2 and COtwo is explained by thermodynamic reasons (Gibbs free free energy) and past bond energy or steric hindrance around the central atom of the molecule.
That is why the more than correct geochemical notation referring to the orthosilicic acid Si(OH)
4 really existing in dilute solution is preferred here. All the same, the main advantage of the now deprecated, but nevertheless ofttimes used, industrial notation referring to the metasilicate anion (SiO 2–
3 ), which also does not exist in aqueous solution, is its greater simplicity and its direct similitude in notation with the carbonate (CO 2–
3 ) organisation.
I will also note that the NaOH and KOH species (alkali hydroxides, besides often simply called alkali to refer to their strongly bones character) which catalyze and accelerate the silica dissolution in the brine-silica reaction practise not explicitly appear in this simplified representation of the ultimate reaction with portlandite, because they are continuously regenerated from the cation exchange reaction with portlandite. As a consequence, they disappear from the global mass rest equation of the catalyzed reaction.
Silica dissolution mechanism [edit]

Anhydrous and hydrated silica surface with siloxane and silanol groups forth with surface jump water molecules.
The surface of solid silica in contact with h2o is covered by siloxane bonds (≡Si–O–Si≡) and silanol groups (≡Si–OH) sensitive to an alkaline attack by OH −
ions.
The presence of these oxygen-bearing groups very prone to class hydrogen bonds with water molecules explains the affinity of silica for water and makes colloidal silica very hydrophile.
Siloxane bonds may undergo hydrolysis and condensation reactions as schematically represented hereafter:

Siloxane bond between 2 silicon atoms.
- ≡Si–O–Si≡ + H
2 O ↔ ≡Si–OH + HO–Si≡ - =Si=O + H
2 O ↔ =Si(–OH)
2
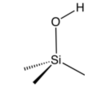
Chemical construction of a silanol group.
On the other paw, silanol groups can too undergo protonation/deprotonation:
- ≡Si–OH ↔ ≡Si–O −
+ H +
.
These equilibria can be shifted towards the right side of the reaction leading to silica dissolution past increasing the concentration of hydroxide anion (OH–), i.eastward., by increasing the pH of the solution.
Alkaline hydrolysis of siloxane bonds occurs past nucleophilic substitution of OH– onto a silicon cantlet, while some other –O–Si group is leaving to preserve the tetravalent character of Si atom:
- ≡Si–O–Si≡ + OH −
→ ≡Si–OH + –O–Si≡ - =Si=O + OH −
→ =SiO(OH) −
Deprotonation of silanol groups:
- ≡Si–OH + OH −
→ ≡Si–O −
+ H
ii O.
In the pH range 0 – 7, the solubility of silica is constant, but to a higher place pH eight, the hydrolysis of siloxane bonds and deprotonation of silanol groups exponentially increment with pH. This is why drinking glass hands dissolves at loftier pH and does non withstand extremely basic NaOH/KOH solutions. Therefore, NaOH/KOH released during cement hydration attacks and dissolves the tridimensional network of silica present in the aggregates. Amorphous or poorly crystallized silica, as cryptocrystalline chalcedony or chert nowadays in flints (in chalk) or rolled river gravels, is much more soluble and sensitive to alkaline attack past OH– anions than well crystallized silica such as quartz. Strained (deformed) quartz or chert exposed to freeze-thaw cycles in Canada and Nordic countries are also more sensitive to alkaline (high pH) solutions.
The species responsible for silica dissolution is the hydroxide anion (OH–). The high pH weather are said to be alkaline and one also speaks of the alkalinity of the basic solutions. For the sake of electroneutrality, (OH–) anions need to be accompanied by positively charged cations, Na+ or Chiliad+ in NaOH or KOH solutions respectively. Na and K both belong to the alkali metals column in the Periodic Table. When speaking of alkali'due south, one systematically refers to NaOH and KOH basic hydroxides, or their respective oxides NaiiO and KiiO in cement. Therefore, information technology is the hydroxide, or the oxide, component of the common salt which is the just relevant chemical species for silica dissolution, not the alkali metallic in itself. Nonetheless, to make up one's mind the alkali equivalent content (NaiiOeq) in cement, considering the need to maintain electroneutrality in solids or in solution, one directly measures the contents of cement in Na and K elements and i conservatively considers that their counter ions are the hydroxide ions. As Na+ and K+ cations are hydrated species, they also contribute to retain water in brine-silica reaction products.
Osmotic processes (Chatterji et al., 1986, 1987, 1989) and the electrical double layer (EDL)[8] play also a primal function in the transport of water towards the concentrated liquid alkali gel explaining their swelling behavior and the deleterious expansion of aggregates responsible of ASR damages in concrete.
Catalysis of ASR past dissolved NaOH or KOH [edit]
The ASR reaction significantly differs from the pozzolanic reaction by the fact that it is catalysed past soluble alkali hydroxides (NaOH / KOH) at very loftier pH. It tin can be represented as follows using the classical geochemical annotation for representing silica by the fully hydrated dissolved silica (Si(OH)4 or silicic acid: HfourSiO4), but an older industrial annotation also exists (H2SiO3, hemihydrated silica (does not exist), by analogy with carbonic acrid):
- 2 Na(OH) + H4SiO4 → Na2H2SiO4 · ii HtwoO
- the so-produced soluble alkali silicagel tin can then react with calcium hydroxide (portlandite) to precipitate insoluble calcium silicate hydrates (C-S-H phases) and regenerate NaOH for continuing the initial silica dissolution reaction:
- Na2HiiSiO4 · 2 HtwoO + Ca(OH)ii → CaHtwoSiO4 · 2 HiiO + two NaOH
The sum, or the combination, of the two above mentioned reactions gives a general reaction resembling the pozzolanic reaction, simply it is important to keep in mind that this reaction is catalysed by the undesirable presence in cement, or other concrete components, of soluble alkaline hydroxydes (NaOH / KOH) responsible for the dissolution of the silica (silicic acid) at high pH:
- Ca(OH)ii + H4SiOfour → CaHiiSiO4 · 2 H2O
Without the presence of dissolved NaOH or KOH, responsible for the loftier pH (~13.v) of the concrete pore h2o, the amorphous silica of the reactive aggregates would not be dissolved and the reaction would not evolve. Moreover, the soluble sodium or potassium silicate is very hygroscopic and swells when information technology absorbs water. When the sodium silicate gel forms and swells inside a porous siliceous aggregate, it commencement expands and occupies the free porosity. When this latter is completely filled, if the soluble merely very viscous gel cannot be easily expelled from the silica network, the hydraulic pressure rises within the attacked amass and leads to its fracture. It is the hydro-mechanical expansion of the damaged siliceous aggregate surrounded by calcium-rich hardened cement paste which is responsible for the development of a network of cracks in concrete. When the sodium silicate expelled from the aggregate encounters grains of portlandite nowadays in the hardened cement paste, an exchange between sodium and calcium cations occurs and hydrated calcium silicate (C-S-H) precipitates with a concomitant release of NaOH. In its turn, the regenerated NaOH can react with the baggy silica aggregate leading to an increased production of soluble sodium silicate. When a continuous rim of C-S-H completely envelops the external surface of the attacked siliceous amass, it behaves every bit a semi-permeable barrier and hinders the expulsion of the sticky sodium silicate while allowing the NaOH / KOH to lengthened from the hardened cement paste within the amass. This selective bulwark of C-Due south-H contributes to increment the hydraulic pressure inside the aggregate and aggravates the cracking procedure. It is the expansion of the aggregates which amercement concrete in the brine-silica reaction.
Portlandite (Ca(OH)2) represents the master reserve of OH– anions in the solid stage as suggested by Davies and Oberholster (1988)[ix] and emphasized by Wang and Gillott (1991).[10] As long as portlandite, or the siliceous aggregates, has not get completely exhausted, the ASR reaction volition continue. The brine hydroxides are continuously regenerated by the reaction of the sodium silicate with portlandite and thus correspond the transmission chugalug of the ASR reaction driving it to completeness. It is thus impossible to interrupt the ASR reaction. The only manner to avoid ASR in the presence of siliceous aggregates and water is to maintain the concentration of soluble brine (NaOH and KOH) at the everyman possible level in physical, so that the catalysis mechanism becomes negligible.
Analogy with the soda lime and concrete carbonatation [edit]
The alkali-silica reaction mechanism catalysed by a soluble stiff base of operations as NaOH or KOH in the presence of Ca(OH)2 (alkalinity buffer present in the solid phase) can exist compared with the carbonatation procedure of soda lime. The silicic acid (H2SiO3 or SiO2) is simply replaced in the reaction by the carbonic acid (HiiCO3 or CO2).
-
(one) CO2 + 2 NaOH → NatwoCOiii + HiiO (COii trapping by soluble NaOH) (2) NaiiCOthree + Ca(OH)2 → CaCOthree + 2 NaOH (regeneration of NaOH later on reaction with lime) sum (1+ii) CO2 + Ca(OH)2 → CaCOthree + H2O (global reaction)
In the presence of water or simply ambience moisture, the strong bases, NaOH or KOH, readily deliquesce in their hydration water (hygroscopic substances, deliquescence phenomenon) and this greatly facilitates the catalysis process because the reaction in aqueous solution occurs much faster than in the dry solid stage.[11] The moist NaOH impregnates the surface and the porosity of calcium hydroxide grains with a high specific surface area.[12] Soda lime is normally used in closed-circuit diving rebreathers and in anesthesia systems.[13] [fourteen]
The same catalytic effect by the alkali hydroxides (function of the Na2Oeq content of cement) also contributes to the carbonatation of portlandite by atmospheric COtwo in concrete although the rate of propagation of the reaction front is there substantially limited by the COtwo diffusion within the physical matrix less porous.[15]
The soda lime carbonatation reaction tin be directly translated into the ancient industrial notation of silicate (referring to the never observed metasilicic acid) but by substituting a C atom by a Si atom in the mass rest equations (i.due east., by replacing a carbonate by a metasilicate anion). This gives the post-obit gear up of reactions likewise commonly encountered in the literature to schematically depict the continuous regeneration of NaOH in ASR:
-
(one) SiO2 + ii NaOH → Na2SiO3 + H2O (SiOii quickly dissolved by hygroscopic NaOH) (2) Na2SiOthree + Ca(OH)2 → CaSiOiii + 2 NaOH (regeneration of NaOH after reaction with portlandite) sum (1+2) SiOtwo + Ca(OH)two → CaSiO3 + HtwoO (global reaction resembling the Pozzolanic reaction)
If NaOH is conspicuously scarce in the system under consideration (soda lime or alkali-silica reaction), it is formally possible to write the same reactions sets by simply replacing the CO3 2- anions by HCO3 − and the SiO3 2- anions by HSiO3 −, the principle of catalysis remaining the same, fifty-fifty if the number of intermediate species differs.
Chief sources of OH −
in hardened cement paste [edit]
Ane can distinguish several sources of hydroxide anions (OH −
) in hardened cement paste (HCP) from the family of Portland cement (pure OPC, with BFS, or with cementitious additions, FA or SF).
Straight sources [edit]
OH −
anions tin be directly nowadays in the HCP pore water or be slowly released from the solid stage (main buffer, or solid stock) by the dissolution of Ca(OH)
2 (portlandite) when its solubility increases when high pH value starts to drib. Beside these ii main sources, ions exchange reactions and precipitation of poorly soluble calcium salts tin can also contribute to release OH −
into solution.
Alkali hydroxides, NaOH and KOH, arise from the directly dissolution of Na
ii O and K
two O oxides produced by the pyrolysis of the raw materials at high temperature (1450 °C) in the cement kiln. The presence of minerals with high Na and K contents in the raw materials tin can thus be problematic. The aboriginal wet manufacturing process of cement, consuming more free energy (water evaporation) that the mod dry process, had the advantage to eliminate much of the soluble Na and K salts present in the raw material.
As previously described in the two sections dealing respectively with ASR catalysis by alkali hydroxides and soda lime carbonatation, soluble NaOH and KOH are continuously regenerated and released into solution when the soluble alkali silicate reacts with Ca(OH)
2 to precipitate insoluble calcium silicate. As suggested by Davies and Oberholster (1988),[ix] the alkali-silica reaction is cocky-perpetuating as the alkali hydroxides are continuously regenerated in the system. Therefore, portlandite is the main buffer of OH −
in the solid stage. As long as the stock of hydroxides in the solid stage is not wearied, the brine-silica reaction can continue to keep until complete disparition of i of the reagents (Ca(OH)
2 or SiO
ii ) involved in the pozzolanic reaction.
Indirect sources [edit]
There exist besides other indirect sources of OH −
, all related to the presence of soluble Na and K salts in the pore water of hardened cement paste (HCP). The start category contains soluble Na and Thousand salts whose corresponding anions can precipitate an insoluble calcium salts, e.g., Na
2 And then
4 , Na
ii CO
iii , Na
3 PO
4 , NaB(OH)
four , Na
2 B
4 O
7 , ... .
Futurity, an example for calcium sulfate (gypsum, anhydrite) precipitation releasing sodium hydroxide:
- Na
ii And so
4 + Ca(OH)
two → CaSO
4 + ii NaOH
or, the reaction of sodium carbonate with portlandite, also important for the catalysis of the brine–carbonate reaction as emphasized past Fournier and Bérubé (2000) and Bérubé et al. (2005):[16] [17]
- Na
two CO
3 + Ca(OH)
two → CaCO
3 + 2 NaOH
Withal, non all Na or K soluble salts can precipitate insoluble calcium salts, such as, e.m., NaCl-based deicing salts:
- 2 NaCl + Ca(OH)
2 ← CaCl
2 + two NaOH
Every bit calcium chloride is a soluble salt, the reaction cannot occur and the chemical equilibrium regresses to the left side of the reaction.
So, a question arises: tin can NaCl or KCl from deicing salts still possibly play a role in the alkali-silica reaction? Na +
and G +
cations in themselves cannot attack silica (the culprit is their counter ion OH −
) and soluble alkali chlorides cannot produce soluble alkali hydroxide by interacting with calcium hydroxide. So, does information technology exist another route to still produce hydroxide anions in the hardened cement paste (HCP)?
Beside portlandite, other hydrated solid phases are present in HCP. The main phases are the calcium silicate hydrates (C-S-H) (the "gum" in cement paste), calcium sulfo-aluminate phases (AFm and AFt, ettringite) and hydrogarnet. C-Southward-H phases are less soluble (~ 10−v M) than portlandite (CH) (~ two.two 10−two Chiliad at 25 °C) and therefore are expected to play a negligible part for the calcium ions release.
An anion-exchange reaction between chloride ions and the hydroxide anions independent in the lattice of some calcium aluminate hydrates (C-A-H), or related phases (C-A-S-H, AFm, AFt), is suspected to as well contribute to the release of hydroxide anions into solution. The principle mechanism is schematically illustrated hereafter for C-A-H phases:
- Cl −
+ (C-A-H)–OH → (C-A-H)–Cl + OH −
Every bit a uncomplicated, but robust, conclusion, the presence of soluble Na and K salts can also cause, past precipitation of poorly soluble calcium salt (with portlandite, CH) or anion exchange reactions (with phases related to C-A-H), the release of OH −
anions into solution. Therefore, the presence of any salts of Na and G in cement pore h2o is undesirable and the measurements of Na and 1000 elements is a good proxy (indicator) for the maximal concentration of OH −
in pore solution. This is why the total brine equivalent content (Na
ii O
eq ) of cement can simply rely on the measurements of Na and K (e.m., by ICP-AES, AAS, XRF measurements techniques).
Alkali gel development and ageing [edit]
The maturation procedure of the fluid alkali silicagel found in exudations into less soluble solid products institute in gel pastes or in efflorescences is described hereafter. Iv singled-out steps are considered in this progressive transformation.[ten]
i. SiO
ii dissolution and Na
2 SiO
three germination (hither, explicitly written in the ancient industrial metasilicate notation (based on the non-existing metasilicic acrid, H
two SiO
3 ) to also illustrate the frequent use of this later in the literature):
- 2 NaOH + SiO
ii → Na
2 SiO
three · H
2 O (immature N-South-H gel) - this reaction is accompanied by hydration and swelling of the alkali gel leading to the expansion of the afflicted aggregates. The pH of the fresh alkali gel is very high and it has often a characteristic amber color. The high pH of immature alkali gel exudations often precludes the growth of mosses at the surface of concrete crack infilling.
2. Maturation of the alkali gel: polymerisation and gelation by the sol–gel process. Condensation of silicate monomers or oligomers dispersed in a colloidal solution (sol) into a biphasic aqueous polymeric network of silicagel. Ca 2+
divalent cations released by calcium hydroxide (portlandite) when the pH starts to slightly drop may influence the gelation process.
3. Cation substitution with calcium hydroxide (portlandite) and precipitation of amorphous calcium silicate hydrates (C-Southward-H) accompanied by NaOH regeneration:
- Na
ii SiO
3 + Ca(OH)
2 → CaSiO
3 + two NaOH
- Amorphous non-stoechiometric calcium silicate hydrates (C-S-H, the not-stoechiometry being denoted here past the utilise of dashes) can recrystallize into rosettes similar to these of gyrolite. The C-South-H formed at this phase tin be considered an evolved calcium silicate hydrate.
4. Carbonation of the C-S-H leading to precipitation of calcium carbonate and amorphous SiO2 stylized as follows:
- CaSiO
3 + CO
2 → CaCO
three + SiO
2
Equally long as the alkali gel (Na
2 SiO
3 ) has non yet reacted with Ca ii+
ions released from portlandite dissolution, it remains fluid and tin can easily exude from broken aggregates or through open cracks in the damage concrete structure. This can lead to visible xanthous viscid liquid exudations (amber liquid droplets) at the surface of affected concrete.
When pH slowly drops due to the progress of the silica dissolution reaction, solubility of calcium hydroxide increases and the alkali gel reacts with Ca 2+
ions. Its viscosity increases due to gelation process and its mobility (fluidity) strongly decreases when C-S-H phases start to precipitate after reaction with calcium hydroxide (portlandite). At this moment, the calcified gel becomes hard, hindering therefore the alkali gel transport in concrete.
When the C-South-H gel is exposed to atmospheric carbon dioxide, information technology undergoes a rapid carbonation and white/yellow efflorescences appear at the surface of concrete. When the relatively fluid alkali gel continue to exude below the hardened superficial gel layer, information technology pushes the efflorescences out of the crevice surface making them to appear in relief. Because the gel drying and carbonation reactions rates are faster than the gel exudation velocity (liquid gel expulsion rate through open cracks), in most of the cases, fresh liquid alkali exudates are not frequently encounterered at the surface of civil engineering concrete structures. Decompressed physical cores can sometimes let observe fresh yellow liquid alkali exudations (mucilaginous amber droplets) only afterward their drilling.
Mechanism of concrete deterioration [edit]
The mechanism of ASR causing the deterioration of concrete can thus be described in four steps as follows:
- The very bones solution (NaOH / KOH) attacks the siliceous aggregates (silicic acid dissolution at high pH), converting the poorly crystallised or amorphous silica to a soluble but very viscous alkali silicate gel (N-S-H, K-Due south-H).
- The consumption of NaOH / KOH past the dissolution reaction of baggy silica decreases the pH of the pore water of the hardened cement paste. This allows the dissolution of Ca(OH)2 (portandite) and increases the concentration of Ca2+ ions into the cement pore water. Calcium ions and then react with the soluble sodium silicate gel to catechumen it into solid calcium silicate hydrates (C-S-H). The C-South-H forms a continuous poorly permeable coating at the external surface of the aggregate.
- The penetrated alkaline solution (NaOH / KOH) converts the remaining siliceous minerals into beefy soluble alkali silicate gel. The resulting expansive force per unit area increases in the core of the amass.
- The accumulated force per unit area cracks the aggregate and the surrounding cement paste when the pressure exceeds the tolerance of the aggregate.[eighteen]
Structural effects of ASR [edit]
The nifty acquired by ASR can have several negative impacts on physical, including:[19]
- Expansion: The swelling nature of ASR gel increases the gamble of expansion in concrete elements.
- Compressive strength: The effect of ASR on compressive forcefulness can be minor for low expansion levels, to relatively higher degrees at larger expansion. (Swamy R.North. 1986) points out that the compressive forcefulness is not very authentic parameter to study the severity of ASR; however, the test is done because of its simplicity.
- Tensile strength / Flexural capacity: Researches show that ASR groovy tin significantly reduce the tensile force of concrete; therefore reducing the flexural capacity of beams. Some research on bridge structures bespeak about 85% loss of capacity equally a result of ASR.
- Modulus of elasticity/UPV: The effect of ASR on rubberband properties of physical and ultrasound pulse velocity (UPV) is very similar to tensile capacity. The modulus of elasticity is shown to be more than sensitive to ASR than pulse velocity.
- Fatigue: ASR reduces the load begetting chapters and the fatigue life of physical (Ahmed T. 2000).
- Shear forcefulness: ASR enhances the shear capacity of reinforced concrete with and without shear reinforcement (Ahmed T. 2000).
Mitigation [edit]
ASR can be mitigated in new concrete by several approaches:
- Limit the alkali metal content of the cement. Many standards[ citation needed ] impose limits on the "Equivalent Na2O" content of cement.
- Limit the reactive silica content of the aggregate. Certain volcanic rocks are particularly susceptible to ASR because they contain volcanic glass (obsidian) and should not be used as aggregate[ commendation needed ]. The employ of calcium carbonate aggregates tin avoid this. In principle, limestone (CaCO3) the level of silica depends on its purity. Some siliceous limestones (a.o., Kieselkalk establish in Switzerland)[20] may be cemented past amorphous or poorly crystalline silica and can be very sensitive to the ASR reaction, as also observed with some Tournaisian siliceous limestones exploited in quarries in the area of Tournai in Kingdom of belgium.[21] The utilise of limestone as aggregate is not a guarantee against ASR in itself. In Canada, the Spratt siliceous limestone is also particularly well known in studies dealing with ASR and is usually used as the Canadian ASR reference amass.
- Add very fine siliceous materials to neutralize the excessive alkalinity of cement with silicic acrid by a controlled pozzolanic reaction at the early on stage of the cement setting. Pozzolanic materials to add together to the mix may be, e.one thousand., pozzolan, silica fume, fly ash, or metakaolin.[22] These react preferentially with the cement alkalis without germination of an expansive pressure, because siliceous minerals in fine particles convert to alkali silicate and then to calcium silicate without formation of semipermeable reaction rims.
- Limit the external alkalis that come in contact with the organization.
A prompt reaction initiated at the early on stage of concrete hardening on very fine silica particles will assistance to suppress a dull and delayed reaction with larger siliceous aggregates on the long term. Following the same principle, the fabrication of depression-pH cement also implies the addition of finely divided pozzolanic materials rich in silicic acid to the concrete mix to decrease its alkalinity. Beside initially lowering the pH value of the physical pore water, the main working mechanism of silica fume addition is to swallow portlandite (the reservoir of hydroxyde (OH–) in the solid phase) and to decrease the porosity of the hardened cement paste past the formation of calcium silicate hydrates (C-South-H). However, silica fume has to be very finely dispersed in the concrete mix because agglomerated flakes of compacted silica smoke can themselves also induce ASR if the dispersion process is bereft. This can exist the case in laboratory studies[ citation needed ] fabricated on cement pastes alone in the absenteeism of aggregates. Silica fume is sufficiently dispersed during mixing operations of big batches of fresh concrete by the presence of coarse and fine aggregates.
As part of a study conducted by the Federal Highway Administration, a diversity of methods accept been applied to field structures suffering from ASR-affected expansion and keen. Some methods, such equally the application of silanes, have shown significant promise, especially when applied to elements such as small columns and highway barriers. The topical application of lithium compounds, have shown piddling or no promise in reducing ASR-induced expansion and great.[23]
Curative treatment [edit]
There are no curative treatments in general for ASR affected structures. Repair in damaged sections is possible, but the reaction will go along. In some cases, when a sufficient drying of thin components (walls, slabs) of a structure is possible, and is followed by the installation of a watertight membrane, the evolution of the reaction can be slowed down, and sometimes stopped, due to the lack of water needed to go along fueling the reaction. Indeed, water plays a triple office in the alkali-silica reaction: solvent for the reaction taking place, transport medium for the dissolved species reacting, and finally besides reagent consumed by the reaction itself.
Nonetheless, physical at the center of thick concrete components or structures tin never dry out considering water transport in saturated or unsaturated weather is always limited by diffusion in the physical pores (water present nether the liquid grade, or nether the vapor state). The h2o diffusion time is thus proportional to the foursquare of its transport distance. Equally a issue, the water saturation degree inside thick concrete structures oftentimes remains college than eighty%, a level sufficient to provide enough water to the arrangement and to maintain the alkali-silica reaction on going.
Massive structures such as dams pose particular problems: they cannot be easily replaced, and the swelling can block spillway gates or turbine operations. Cutting slots across the construction can relieve some pressure, and help restore geometry and part.
Heavy aggregates for nuclear shielding concrete [edit]
Ii types of heavy aggregates are commonly used for nuclear shielding physical in order to efficiently absorb gamma-rays: baryte (BaSO
4 , density = 4.3 – four.5) and diverse types of atomic number 26 oxides, mainly magnetite (Fe
3 O
4 , density = 5.2) and hematite (Fe
ii O
3 , density = five.iii). The reason is their high density favorable to gamma attenuation. Both types of aggregates need to be checked for ASR as they may contain reactive silica impurities nether a class or another.[24] [25] [26]
As elevated temperature in the range of 50 – lxxx °C may be reached in the concrete of the primary confinement wall around nuclear reactors, particular attending has to exist paid to the pick of aggregates and heavy aggregates to avoid alkali-silica reaction promoted by reactive silica impurities and accelerated by the high temperature to which concrete is exposed.
In some hydrothermal deposits, baryte is associated with silica mineralization[27] and tin can also contain reactive cristobalite[28] [29] while oxy-hydroxides of Fe(3), in particular ferrihydrite, showroom a potent analogousness for dissolved silica nowadays in h2o and may establish an ultimate sink for SiO
2 .[xxx]
This explains how microcrystalline silica tin progressively accumulate in the mineral gangue of iron oxides.[31]
Dissolved silica (Si(OH)
iv ), and its corresponding silicate anion (SiO(OH) −
3 ), strongly sorbs onto hydrous ferric oxides (HFO) and ferric oxides hydrated surface (>Atomic number 26–OH) by ligand exchange:
- (HO)
3 SiO −
+ >Fe–OH → >Fe–OSi(OH)
three + OH −
In this ligand exchange reaction, a silicate anion (also oftentimes more simply written every bit H
iii SiO −
4 ) is making a nucleophilic commutation onto a >Atomic number 26–OH ferrol surface group of HFO and ejects a hydroxide anion OH −
while taking its place onto the ferrol group. This mechanism explains the formation of strong inner sphere complexes of silica at the surface of fe oxy-hydroxides and iron oxides.[32] The surface of iron oxides becomes progressively coated with silica and a silica gangue forms at the surface of iron oxide ores. This explains why some iron ores are rich in silica and may therefore exist sensitive to the brine-silica reaction. Very low level of reactive silica in heavy aggregates are sufficient to induce ASR. This is why heavy aggregates must be systematically tested for ASR before nuclear applications such as radiation shielding or immobilization of strongly irradiating nuclear waste.
Some other reason of concern for the possible accelerated evolution of ASR in the concrete of nuclear structures is the progressive amorphization of the silica independent in aggregates exposed to high neutron fluence.[33] This process is also known every bit metamictization and is known to create amorphous halo's in minerals like zircon rich in uranium and thorium when their crystal construction is submitted to intense alpha-particles internal bombardment and becomes amorph (metamict country).
The loss of mechanical properties of heavily neutron-irradiated concrete component such every bit the biological shield of a reactor at the terminate of the service life of a nuclear power plant is expected to be due to radiation-induced swelling of aggregates, which leads to volumetric expansion of the concrete.[34]
Prevention of the risk [edit]
The just way to prevent, or to limit, the gamble of ASR is to avert one or several of the 3 elements in the disquisitional triangle amass reactivity – cement alkali content – h2o:
- by selecting not-reactive aggregates subsequently testing them according to an appropriate standard test method (see next section);
- by using a low-alkali (LA) cement: with a maximum brine content expressed in Na
2 O
eq < 0.60% of the cement mass, according to EN 197-one European standard for cement,[35] or by limiting the full alkali content in concrete (e.yard., less than 3 kg Na
ii O
eq /m3 of concrete for a CEM I cement (OPC)). Example of standard for concrete in Belgium: NBN EN 206 and its national supplement NBN B 15-001;[36] [37] - by limiting the contact of underground or meteoritic water infiltrations with the concrete structure (h2o tight membrane, roofing, sufficient water drainage, ...). This concluding precaution is always advisable when possible and the only one also sometimes applicable for existing ASR-affected physical structures.
Methods for testing potential alkali reactivity [edit]
The American Lodge for Testing and Materials (ASTM International) has adult dissimilar standardized test methods for screening aggregates for their susceptibility to ASR:
- ASTM C227: "Exam Method for Potential Alkali Reactivity of Cement-Aggregate Combinations (Mortar-Bar Method)"
- ASTM C289: "Standard Test Method for Potential Brine-Silica Reactivity of Aggregates (Chemical Method)"
- ASTM C295: "Guide for Petrographic Examination of Aggregate for Concrete"
- ASTM C1260: "Test Method for Potential Reactivity of Aggregates (Mortar-Bar-Test)". It is a rapid examination of aggregates: immersion of mortar bars in NaOH 1 M at lxxx °C for fourteen days used to apace place highly reactive aggregates or quasi non-reactive aggregates. Beside an elevated temperature, the C1260 method also involves the apply of a large quantity/inventory of NaOH in the solution in which the mortar bar is immersed. A big pool of OH– anions is thus available to diffuse inside the mortar bar to dissolve silica present in aggregates. Consequently, this test is very severe and may exclude valuable aggregates. In case of not-decisive results, the long-term ASTM C1293 test method has to be used for a terminal screening. The main advantage of the ASTM C1260 examination is that it allows to quickly identify extreme cases: very insensitive or very reactive aggregates.
- ASTM C1293: "Test Method for Physical Aggregates by Decision of Length Change of Concrete Due to Brine-Silica Reaction". It is a long-term confirmation test (1 or two years) at 38 °C in a water-saturated moist atmosphere (within a thermostated oven) with concrete prisms containing the aggregates to exist characterised mixed with a high-alkali cement specially selected to induce ASR. The concrete prisms are not straight immersed in an alkaline solution, but wrapped with moist tissues and tightly packed inside a water-tight plastic foils.
- ASTM C1567: "Standard Test Method for Determining the Potential Brine-Silica Reactivity of Combinations of Cementitious Materials and Amass (Accelerated Mortar-Bar Method)"
Other concrete prism methods have also been internationally adult to notice potential alkali-reactivity of aggregates or sometimes hardened concrete cores, east.chiliad.:
- The Oberholster method on which the ASTM C1260 test is based. It is a astringent short duration test with immersion of the mortar prism or physical cadre in a solution of NaOH 1 M at eighty °C for 14 days.[38]
- The Duggan method starts with a offset immersion of several concrete cores in distilled water at 22 °C for rehydration during 3 days. It is then followed by heating for one twenty-four hour period in a dry out oven at 82 °C and then with a succession of cycles of one twenty-four hours hydration followed by one solar day drying at 82 °C. The expansion of the concrete cores is measured till 14 or 20 days.[39] [40] [41] [42] It is a short elapsing test for ASR/AAR but much softer than the Oberholster exam. Information technology can too be used to measure the expansion of concrete due to delayed ettringite germination (DEF). The mechanical stresses induced by the thermal cycles create micro-cracks in the concrete matrix and then facilitate the accessibility to h2o of the reactive mineral phases in the treated samples.[43]
- The concrete microbar examination was proposed by Grattan-Bellew et al. (2003) as a universal accelerated test for alkali-aggregate reaction.[44]
- CSA A23.1-14A and CSA A23.two-14A: Canadian CSA standard physical prism tests for potential expansivity of cement/aggregate combinations.[45] [43] CSA A23.two-14A is a long-term test in which concrete prisms are stored under saturated moist weather condition at a temperature of 38 °C, for a minimum of 365 days. It is the Canadian standard equivalent to ASTM C1293.
- LCPC/IFSTTAR (1997) LPC-44. Brine reaction in concrete. Residual expansion tests on hardened physical.[46]
- RILEM AAR-3 concrete prism method (storage at 38 °C).
- RILEM AAR-4 concrete prism method (storage at lx °C).
- RILEM AAR-4 alternative method (storage at 60 °C).
- German concrete test method (storage at twoscore °C).
- Norwegian physical prism method (storage at 38 °C).
Known affected structures [edit]

Surface of a physical pillar of the edifice of the National Gallery of Canada at Ottawa presenting the typical cleft pattern of the alkali-silica reaction (ASR). Note the typical fatty aspect of the xanthous silicagel exudations imbibing the concrete surface along the two sides of the cracks.
Australia [edit]
- Adelaide Festival Middle car park, demolished in 2017[47]
- Centennial Hall, Adelaide (1936-2007)
- Dee Why bounding main puddle, Dee Why, Australia.[48]
- Rex St Bridge, demolished and replaced in 2011 (crossing the Patawalonga River, Glenelg North, South Australia).[49] [l]
- Manly Surf Pavilion, Manly, Australia (1939–1981).[51]
- The MCG'south old Southern Stand, demolished in 1990 and replaced with the Great Southern Stand up which was completed in 1992
- Westpoint Blacktown car park
Belgium [edit]
- Many bridges and civil engineering works of motorways because the improper apply of highly reactive siliceous Tournaisian limestone (lower carboniferous Dinantian) during the years 1960 – 1970 when most of the motorways were constructed in Belgium. ASR damages started to be recognised only in the 1980s.[52] The Tournaisian limestone may comprise up to 25 – 30 wt. % of reactive biogenic silica originating from the spicules of siliceous sponges deposited with calcium carbonate in the marine sediments.
- Pommeroeul lock in Hainaut on the canal Hensies – Pommeroeul – Condé.
- Tour & Taxis auto admission ramp in Brussels with liquid exudations of bister alkali gel evidenced on concrete cores past SPW experts (Public Services of Wallonia).
- External containment building of the Tihange 2 nuclear ability plant.
- Poorly conditioned radioactive waste from the Doel nuclear power plant: evaporator concentrates and spent ion-substitution resins (SIER) exuding out of the concrete immobilization matrix very large quantities of liquid sodium silicagel.[53] [54] [55] [56] [57] [58]
Canada [edit]
Brine-aggregate reactions (AAR), both brine-silica (ASR) and brine-carbonate (ACR, involving dolomite) reactions, were identified in Canada since the years 1950'due south.[59] [threescore] [61]
- Many hydraulic dams are affected past ASR in Canada because of the wide use of reactive aggregates.[62] Indeed, reactive frost-sensitive chert is very ofttimes plant in glacio-fluvial environments from which gravels are ordinarily extracted in Canada. Some other reason is also the presence of reactive silica in Paleozoic limestones similar the siliceous Ordovician limestone (Bobcaygeon Formation) from the Spratt's quarry almost Ottawa in Ontario.[63] The Spratt's limestone aggregates (from the visitor "Spratt Sand and Gravel Express") are widely used for ASR studies in Canada and worldwide as described by Rogers et al. (2000)[59] and also recommended by RILEM (International Matrimony of Laboratories and Experts in Structure Materials, Systems, and Structures).[64]
- Many bridges and ceremonious engineering works of motorways.
- Interchange Robert Bourassa – Charest (Québec city: interchange autoroutes 740 – 440) demolished in 2010.[3]
- Gentilly 2 nuclear power plant.
- Building of the National Gallery of Canada at Ottawa.
France [edit]
- Former Térénez bridge in Brittany, built in 1951 and replaced in 2011.
Frg [edit]
- East German Deutsche Reichsbahn used numerous physical ties in the 1970s to supervene upon previous wooden ties. However, the gravel from the Baltic Sea acquired ASR and the ties had to be replaced earlier than planned, lasting well into the 1990s.[ citation needed ]
- Afterward reunification, many Autobahns in East Frg were refurbished with physical that turned out to have been defective and affected by ASR, necessitating expensive replacement work.[ citation needed ]
New Zealand [edit]
- Fairfield Bridge in Hamilton, New Zealand. Repaired in 1991 at a price of NZ$1.1 meg.[65]
U.k. [edit]
- Keybridge House,[66] Southward Lambeth Road, Vauxhall, London, England.
- Millennium Stadium N Stand (part of the sometime National Stadium),[67] Cardiff, Wales.
- Merafield Span,[68] A38, England. Demolished manually in 2016.[69]
- Pebble Mill Studios, Birmingham. Demolished in 2005 [70]
- Imperial Devon and Exeter Hospital, Wonford. Demolished and replaced in the mid-1990s.[71] [72]
- Steve Bull Stand, Molineux Stadium, Wolverhampton
United States [edit]
- Seabrook Station Nuclear Power Plant in Seabrook, New Hampshire.
- Seminoe Dam in Wyoming.[73]
- Chickamauga Dam in Tennessee.[74]
- Sixth Street Viaduct in Los Angeles. Demolished in 2016.
- References for ASR very reactive aggregates in the United states:
- Coarse aggregates: volcanic rock from New Mexico
- Fine aggregates: siliceous sand from Texas
See too [edit]
- Brine-carbonate reaction
- Alkali–aggregate reaction
- Calthemite: Secondary calcium carbonate eolith growing under man-fabricated structures
- Carbonatation
- Colloidal silica
- Structure aggregate
- Keen pattern
- Crocodile cracking: distress in asphalt pavement characterized by interconnecting or interlaced cracking in the asphalt layer
- Energetically modified cement (EMC)
- Gyrolite, a product of slag hydration and ASR gel ageing
- Hydrated silica
- Pozzolanic reaction
- Silicate: come across solid SiO2 hydrolysis/dissolution and Si–OH deprotonation reactions at high pH
- Siliceous sponge
- Soda lime: the mechanism of ASR catalysed by NaOH is coordinating to the trapping mechanism of COtwo past Ca(OH)2 impregnated with NaOH
External links [edit]
- Understanding cement website treatise on ASR
- PCA treatise on ASR
- Concrete Construction Net treatise of ASR
- The states Federal Highway Administration (FHWA) treatise on the apply of lithium to foreclose or mitigate ASR
- Clan of German Cement Works – Alkali-silica reaction – Overview
Reference textbooks [edit]
- Sims, Ian; Poole, Alan B. (2017-08-01). Brine-aggregate reaction in concrete: A world review. CRC Press. ISBN978-1-317-48441-7.
References [edit]
- ^ FHWA (2010-06-22). "Alkali-Silica Reactivity (ASR) – Concrete – Pavements – FHWA". Alkali-Silica Reactivity (ASR) Development and Deployment Program. Archived from the original on viii August 2010. Retrieved 2010-07-28 .
- ^ Faridazar, Fred (2009-02-10). "Techbrief: Selecting candidate structures for lithium handling: What to provide the petrographer along with concrete specimens, FHWA-HRT-06-069 – Pavements – FHWA". FHWA-HRT-06-069 . Retrieved 2010-07-28 .
- ^ a b Fournier, B., Sanchez, 50., & Beauchemin, S. (2015). Outils d'investigation de la réactivité alcalis-granulats dans les infrastructures en béton. Rapport Final, Ministère des transports du Québec, Service des matériaux d'infrastructure, Secteur béton de ciment, août (Vol. 2015, p. 293).
- ^ "Alkali–silica reaction in concrete". Understanding Cement. Archived from the original on 10 Baronial 2007. Retrieved 2007-08-11 .
- ^ "Merafield Bridge in Plympton demolished". BBC News . Retrieved 2016-05-16 .
- ^ Stanton, T.E. (1940). "Expansion of concrete through reaction between cement and aggregate". Engineering News-Tape. No. SP-249-1.
- ^ Wigum, B.J.; Pedersen, 50.T.; Grelk, B.; Lindgard, J. (2006). Country-of-the fine art report: Cardinal parameters influencing the alkali aggregate reaction. SBF52 A06018 — Unrestricted Report. 134 pp (PDF). SINTEF Edifice and Infrastructure. ISBN82-14-04078-7.
- ^ Prezzi, Monica; Monteiro, Paulo J.M.; Sposito, Garrison (1997). "The alkali–silica reaction: Part I. Utilize of the double-layer theory to explain the behavior of reaction-product gels". ACI Materials Journal. 94 (i): 10–17. ISSN 0889-325X.
- ^ a b Davies, Yard.; Oberholster, R. E. (1988-07-01). "Alkali-silica reaction products and their development". Cement and Physical Research. xviii (four): 621–635. doi:10.1016/0008-8846(88)90055-5. ISSN 0008-8846. Retrieved 2020-10-31 .
- ^ a b Wang, H.; Gillott, J. East. (1991-07-01). "Machinery of brine-silica reaction and the significance of calcium hydroxide". Cement and Concrete Research. 21 (4): 647–654. doi:10.1016/0008-8846(91)90115-X. ISSN 0008-8846.
- ^ Samari, Mohammad; Ridha, Firas; Manovic, Vasilije; Macchi, Arturo; Anthony, E. J. (2019). "Direct capture of carbon dioxide from air via lime-based sorbents". Mitigation and Adaptation Strategies for Global Modify. 25: 25–41. doi:10.1007/s11027-019-9845-0. ISSN 1381-2386.
- ^ Ševčík, Radek; Mácová, Petra; Sotiriadis, Konstantinos; Pérez-Estébanez, Marta; Viani, Alberto; Šašek, Petr (2016). "Micro-Raman spectroscopy investigation of the carbonation reaction in a lime paste produced with a traditional technology". Journal of Raman Spectroscopy. 47 (12): 1452–1457. Bibcode:2016JRSp...47.1452S. doi:10.1002/jrs.4929. ISSN 0377-0486.
- ^ Adriani, J.; Byrd, Yard. L. (1941). "A study of carbon dioxide absorption appliances for anesthesia: The canister". Anesthesiology: The Periodical of the American Society of Anesthesiologists. 2 (four): 450–455.
- ^ Freeman, Brian S.; Berger, Jeffrey S. (2014). Anesthesiology Core Review: Part One Bones Exam. Chapter 17: Absorption of Carbon Dioxide. McGraw-Hill Education. Retrieved 22 April 2020 – via Access Medicine.
- ^ Verbeck, Thou. (1958). "Carbonation of hydrated Portland Cement". STP205-EB Cement and Concrete (W Conshohocken, PA: ASTM International: 17–36. doi:10.1520/STP39460S. ISBN978-0-8031-5667-viii.
- ^ Fournier, B., & Bérubé, M. A. (2000). Alkali-aggregate reaction in physical: a review of basic concepts and engineering implications. Canadian Journal of Civil Engineering, 27(2), 167-191. Run into the chemic equations on p. 168.
- ^ Bérubé, M. A., Smaoui, Due north., Bissonnette, B., & Fournier, B. (2005). Outil d'évaluation et de gestion des ouvrages d'art affectés de réactions alcalis-silice (RAS). Études et Recherches en Transport, Ministère des Transports du Québec. See the chemical equations on pp. iii-4.
- ^ Ichikawa, T.; Miura, M. (2007). "Modified model of alkali-silica reaction". Cement and Concrete Research. 37 (9): 1291–1297. doi:10.1016/j.cemconres.2007.06.008.
- ^ "Structural Effects of ASR on Concrete Structures | FPrimeC Solutions". FPrimeC Solutions. 2016-10-28. Retrieved 2017-01-11 .
- ^ Funk, Hanspeter (1975). "The origin of authigenic quartz in the Helvetic Siliceous Limestone (Helvetischer Kieselkalk), Switzerland". Sedimentology. 22 (2): 299–306. Bibcode:1975Sedim..22..299F. doi:ten.1111/j.1365-3091.1975.tb00296.x.
- ^ Monnin, Y.; Dégrugilliers P.; Bulteel D.; Garcia-Diaz E. (2006). "Petrography study of two siliceous limestones submitted to alkali-silica reaction". Cement and Concrete Inquiry. 36 (8): 1460–1466. doi:ten.1016/j.cemconres.2006.03.025. ISSN 0008-8846.
- ^ Ramlochan, Terrence; Michael Thomas; Karen A. Gruber (2000). "The result of metakaolin on alkali-silica reaction in physical". Cement and Physical Research. thirty (three): 339–344. doi:10.1016/S0008-8846(99)00261-6. ISSN 0008-8846.
- ^ "Publication Details for Alkali-Aggregate Reactivity (AAR) Facts Book - Pavements - FHWA". dot.gov.
- ^ Jóźwiak-Niedźwiedzka, Daria; Gibas, Karolina; Brandt, Andrzej Yard.; Glinicki, Michał A.; Dąbrowski, Mariusz; Denis, Piotr (2015). "Mineral composition of heavy aggregates for nuclear shielding concrete in relation to alkali-silica reaction". Procedia Engineering. 108: 162–169. doi:x.1016/j.proeng.2015.06.132. ISSN 1877-7058.
- ^ Jóźwiak-Niedźwiedzka, Daria; Glinicki, Michał A.; Gibas, Glinicki (2016). Potential for alkali–silica reaction in radiations shielding physical containing special aggregates. 5th International Conference on the Durability of Physical Structures. Purdue University Press. doi:10.5703/1288284316137. ISBN978-i-62671-065-8 . Retrieved 2021-06-14 .
- ^ Jóźwiak-Niedźwiedzka, Daria; Glinicki, Michał; Gibas, Karolina; Baran, Tomasz (2018). "Alkali-silica reactivity of high density aggregates for radiation shielding concrete". Materials. eleven (xi): 2284. doi:10.3390/ma11112284. ISSN 1996-1944. PMC6265780.
- ^ Fedele, L.; Todesca, R.; Boni, Yard. (2003). "Barite-silica mineralization at the inter-Ordovician unconformity in southwestern Sardinia (Italia): a fluid inclusion study". Mineralogy and Petrology. 77 (iii–four): 197–213. Bibcode:2003MinPe..77..197F. doi:10.1007/s00710-002-0200-9. ISSN 0930-0708. S2CID 129874363.
- ^ Jóźwiak-Niedźwiedzka, Daria; Brandt, Andrzej; Gibas, Karolina; Denis, Piotr (2014). "The alkali-amass reaction gamble in the case of barite concretes". Cement, Wapno, Beton. 19: 234–242.
- ^ Jówiak-Niedźwiedzka, D.; Brandt, Andrzej; Gibas, Karolina; Denis, Piotr (2014). Influence of barite composition on potential alkali aggregate reaction in radiations-shielding physical. 2d Briefing on Technological Innovations in Nuclear Civil Applied science TINCE 2014, Paris, 1st to quaternary September 2014. Vol. 2014. Paris. pp. 234–242.
- ^ Carlson, 50.; Schwertmann, U. (1981). "Natural ferrihydrites in surface deposits from Finland and their association with silica". Geochimica et Cosmochimica Acta. 45 (3): 421–429. doi:x.1016/0016-7037(81)90250-vii. ISSN 0016-7037.
- ^ Taylor, P. (1995). Interactions of silica with iron oxides: Furnishings on oxide transformations and sorption properties. Diminutive Energy of Canada Ltd. (AECL). Retrieved 2021-06-xvi .
- ^ Jordan, Norbert; Marmier, Nicolas; Lomenech, Claire; Giffaut, Eric; Ehrhardt, Jean-Jacques (2007). "Sorption of silicates on goethite, hematite, and magnetite: Experiments and modelling". Journal of Colloid and Interface Science. 312 (2): 224–229. doi:10.1016/j.jcis.2007.03.053. ISSN 0021-9797.
- ^ Rosseel, Thomas G.; Maruyama, Ippei; Le Pape, Yann; Kontani, Osamu; Giorla, Alain B.; Remec, Igor; Wall, James J.; Sircar, Madhumita; Andrade, Carmen; Ordonez, Manuel (2016). "Review of the current state of cognition on the effects of radiation on concrete". Journal of Advanced Concrete Engineering. 14 (seven): 368–383. doi:x.3151/jact.xiv.368. ISSN 1346-8014.
- ^ Field, Chiliad.1000.; Remec, I.; Pape, Y. Le (2015). "Radiation effects in concrete for nuclear ability plants – Office I: Quantification of radiation exposure and radiation effects". Nuclear Engineering and Design. 282: 126–143. doi:10.1016/j.nucengdes.2014.x.003. ISSN 0029-5493.
- ^ EN 197-1 European Standard. Cement – Part ane: Composition, specifications and conformity criteria for common cements.
- ^ NBN EN 206:2013+A1:2016 Concrete – Specification, performance, product and conformity. Publication date: 11/2016.
- ^ NBN B fifteen-001:2018. Concrete – Specification, performance, production and conformity – National supplement for Belgium to NBN EN 206:2013+A1:2016. Publication date: 07/2018.
- ^ Oberholster, R. E.; Davies, G. (1986). "An accelerated method for testing the potential brine reactivity of siliceous aggregates". Cement and Concrete Research. 16 (2): 181–189. doi:10.1016/0008-8846(86)90134-1. ISSN 0008-8846. Retrieved 2020-10-25 .
- ^ Scott, J.F., Duggan, C.R., (1986). Potential new test for alkali amass reactivity, Roe. 7th Intl. Conf. on Brine Aggregate Reactions, Ottawa Canada, ed. P.Due east. Grattan-Bellew, Noyes publ., North.J., USA, 319-323.
- ^ Duggan C.R., Scott J.F. (1987). Proposed new examination for alkali-aggregate reactivity, Canadian National Railways, Technical Research Report, Montreal, Canada, Apr 13, 1987, revised Oct. 31,1989.
- ^ Duggan C.R. and Scott J.F. (1989a). Institution of new acceptance rejection limits for proposed test method for detection of potentially deleterious expansion of concrete, presented to ASTM Subcommittee C09.02.02, sept 1989.
- ^ Duggan C.R. and Scott J.F. (1989b). New examination for deleterious expansion in concrete, eighth Intl. Conf. on Alkali-Aggregate Reaction Kyoto, Japan, 403408.
- ^ a b Mean solar day, R. 50. (1992). The effect of secondary ettringite germination on the immovability of concrete: A literature assay (No. RD108T). See mainly Affiliate vii: Rapid test method for secondary ettringite formation. pp. 81-95 of the PDF file (pp. 69-83 of the hard copy). Available in open access on the site of Cement.org
- ^ Grattan-Bellew, P.E.; G. Cybanski; B. Fournier; 50. Mitchell (2003). "Proposed universal accelerated test for brine-aggregate reaction: the concrete microbar test". Cement Concrete and Aggregates. 25 (2): 29–34.
- ^ A23.1-14/A23.2-14 Concrete materials and methods of physical construction / Test methods and standard practices for physical. Published by CSA Group in 2014, 690 pages.
- ^ LCPC/IFSTTAR (1997) Alcali-réaction du béton. Essai d'expansion résiduelle sur béton durci. Projet de méthode d'essai LCP 44. Février 1997. 15 pp. MethodeDEssai-LCPC-ME44.pdf. https://www.ifsttar.fr/fileadmin/user_upload/editions/lcpc/MethodeDEssai/MethodeDEssai-LCPC-ME44.pdf
- ^ "Adelaide Festival Plaza redevelopment".
- ^ "Manly Daily | News Local Paper | Daily Telegraph | Manly Daily | Daily Telegraph".
- ^ Anna Vlach, The Adelaide Advertiser, "Pat bridge load fears", 8 Baronial 2007, page 9.
- ^ Jane Whitford Guardian Messenger December 14, 2011
- ^ "404". world wide web.architecture.com.au.
- ^ "ASR first recognised in Belgium civil engineering science structures — Service Public Wallon: Management de 50'expertise des ouvrages" (PDF). 2010-09-15.
- ^ http://www.ondraf.exist/, web site of ONDRAF/NIRAS announcing in September 2013 the discovery on the interim storage site of Belgoprocess at Dessel of ten 000 LILW drums afflicted or potentially affected by ASR.
- ^ "MONA website: Conditioned radioactive waste afflicted past ASR in Belgium — Gelvaten niet geschikt voor berging" (PDF). 2014-03-01. [ permanent dead link ]
- ^ "STORA website: Conditioned radioactive waste afflicted by ASR in Belgium — Gelvaten problematiek".
- ^ "STORA website: Conditioned radioactive waste afflicted by ASR in Belgium — Nieuw opslaggebouw voor gelvaten". 2014-09-26.
- ^ "STORA website: Conditioned radioactive waste affected by ASR in Belgium — Nieuw opslaggebouw voor gelvaten klaar in 2019". 2016-09-29.
- ^ "NIRAS magazine June 2016: Conditioned radioactive waste affected past ASR in Belgium — Nieuw hoogtechnologisch opslaggebouw voor vaten met gelvorming, pp. twenty-21" (PDF). 2016-06-01.
- ^ a b Rogers, C., Grattan-Bellew, P. East., Hooton, R. D., Ryell, J., & Thomas, M. D. (2000). Brine-aggregate reactions in Ontario. Canadian Journal of Civil Applied science, 27(2), 246-260.
- ^ Fournier, B., & Bérubé, Thou. A. (2000). Alkali-aggregate reaction in concrete: a review of basic concepts and engineering implications. Canadian Journal of Civil Engineering science, 27(2), 167-191.
- ^ Bérubé, M. A., Smaoui, N., Bissonnette, B., & Fournier, B. (2005). Outil d'évaluation et de gestion des ouvrages d'art affectés de réactions alcalis-silice (RAS). Études et Recherches en Transport, Ministère des Transports du Québec.
- ^ Du, Chongjiang (2010). "HRW-Hydro Review Worldwide: Dealing with brine-aggregate reaction in hydraulic structures".
- ^ Limestone Industries of Ontario, Volume 2 Ontario Geological Survey. Engineering science and Terrain Geology Section Ontario, Ministry building of Natural Resources, 1989 – Limestone – 196 pages
- ^ Nixon, J. P., & Sims, I. (Eds.). (2016). RILEM recommendation of the prevention of damage by alkali-aggregate reactions in new concrete structures. Dordrecht: Springer.
- ^ "Fairfield Bridge". Hamilton City Libraries. Archived from the original on 2009-10-23. Retrieved 2009-ten-23 .
- ^ "Keybridge Business firm, London - Building #1458". www.skyscrapernews.com.
- ^ Laura Kemp (viii July 2007) "The Millennium Stadium is suffering from concrete cancer, we can reveal", Wales on Dominicus.
- ^ "A38 Merafield Bridge replacement". www.highways.gov.uk. 27 Baronial 2014. Archived from the original on 21 May 2016. Retrieved 16 May 2016.
- ^ "Merafield Bridge in Plympton demolished". BBC News. 16 May 2016. Retrieved 14 August 2017.
- ^ "Pebble Mill Studios". BUFVC. BUFC. Retrieved 27 March 2018.
- ^ Warner, Brian (26 March 1992). "UK: Eight contractors prepare bids for £22m Royal Devon and Exeter Hospital redevelopment contract". Construction News.
- ^ "The Royal Devon and Exeter Infirmary". Exeter Memories. 7 January 2014.
- ^ Written report DSO-2014-03: Seminoe dam – Assessment of concrete past quantitative methods – The petrographic damage rating index.
- ^ "Tennessee Valley Authority (TVA) to have a slice out of Chickamauga Dam". www.chattanoogan.com . Retrieved 2020-xi-10 .
Source: https://en.wikipedia.org/wiki/Alkali%E2%80%93silica_reaction
0 Response to "How to Deduce the Alkali Metal When You Know the Anion Is Carbonate"
Post a Comment